Abstract
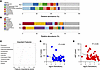
Human islet antigen reactive CD4+ memory T cells (IAR T cells) play a key role in the pathogenesis of autoimmune type 1 diabetes (T1D). Using single-cell RNA sequencing (scRNA-Seq) to identify T cell receptors (TCRs) in IAR T cells, we have identified a class of TCRs that share TCRα chains between individuals (“public” chains). We isolated IAR T cells from blood of healthy, new-onset T1D and established T1D donors using multiplexed CD154 enrichment and identified paired TCRα/β sequences from 2767 individual cells. More than a quarter of cells shared TCR junctions between 2 or more cells (“expanded”), and 29/47 (~62%) of expanded TCRs tested showed specificity for islet antigen epitopes. Public TCRs sharing TCRα junctions were most prominent in new-onset T1D. Public TCR sequences were more germline like than expanded unique, or “private,” TCRs, and had shorter junction sequences, suggestive of fewer random nucleotide insertions. Public TCRα junctions were often paired with mismatched TCRβ junctions in TCRs; remarkably, a subset of these TCRs exhibited cross-reactivity toward distinct islet antigen peptides. Our findings demonstrate a prevalent population of IAR T cells with diverse specificities determined by TCRs with restricted TCRα junctions and germline-constrained antigen recognition properties. Since these “innate-like” TCRs differ from previously described immunodominant TCRβ chains in autoimmunity, they have implications for fundamental studies of disease mechanisms. Self-reactive restricted TCRα chains and their associated epitopes should be considered in fundamental and translational investigations of TCRs in T1D.
Authors
Peter S. Linsley, Fariba Barahmand-pour-Whitman, Elisa Balmas, Hannah A. DeBerg, Kaitlin J. Flynn, Alex K. Hu, Mario G. Rosasco, Janice Chen, Colin O’Rourke, Elisavet Serti, Vivian H. Gersuk, Keshav Motwani, Howard R. Seay, Todd M. Brusko, William W. Kwok, Cate Speake, Carla J. Greenbaum, Gerald T. Nepom, Karen Cerosaletti
Abstract
Metabolic dysfunction–associated steatotic liver disease (MASLD) is a global health concern with limited interventions. While the role of gut bacteria in MASLD has been extensively studied, the contribution of gut fungi remains largely unexplored. This study investigates the impact of fungal dysbiosis and the role of CARD9, a key adaptor protein in fungal sensing on gut-liver axis dysfunction in MASLD. Patients with advanced liver fibrosis exhibited distinct mycobiota profiles. Using a Card9-deficient mouse model subjected to high-fat, high-glucose/-fructose feeding, we observed exacerbated liver injury and fibrosis accompanied by fungal dysbiosis, paralleling our findings in human patients. Beyond its established expression in myeloid cells, CARD9 was also detected in intestinal enterocytes where its expression was diminished under metabolic stress. Intestinal organoids with CARD9 inhibition had reduced expression of antimicrobial Reg3g, the tight junction protein ZO-1, and the antifungal enteroendocrine hormone PYY. These findings suggest that CARD9 maintains gut barrier integrity, preventing microbial translocation and subsequent liver injury and fibrosis. Our results provide insights into the interplay between fungal dysbiosis, gut barrier dysfunction, and MASLD, and identify CARD9 as a key protein within this axis.
Authors
Vijay Pandyarajan, So Yeon Kim, Takashi Tsuchiya, Selena Liu, Sadam H. Bhat, Jieun Kim, David M. Underhill, Mazen Noureddin, Shelly C. Lu, Ekihiro Seki
Abstract
Patients with COVID-19 who develop platelet-activating antibodies represent a subset at heightened thrombotic risk, yet the immune features associated with this response remains to be defined. We applied single-cell RNA-seq of B and T cells, single B cell V(D)J-seq, and plasma cytokine and chemokine analysis to define immune signatures distinguishing patients who did (PEA+) or did not (PEA–) develop these antibodies. Patients positive for PEA showed prominent transcriptional enrichment of inflammatory, antigen presentation, and B cell receptor signaling pathways within antigen-experienced B cell subsets. Expanded B cell clones in patients positive for PEA were disproportionately enriched within atypical memory B cells and exhibited upregulated IFN-γ–response signatures, increased proliferative mutational patterns, limited class switching, and a significant overrepresentation of RKH/Y5 heavy-chain motifs associated with platelet-activating antibodies, consistent with an extrafollicular-biased response. Parallel T cell profiling revealed IL-12 pathway enrichment across most T cell subsets, increased IFN-γ transcription, and elevated plasma levels of Th1-associated cytokines in patients positive for PEA. Collectively, these data highlight a coordinated inflammatory environment marked by Th1-skewed T cell activation and selective expansion of atypical memory B cell clones carrying RKH/Y5 motifs, defining immunologic features associated with platelet-activating antibody development in COVID-19.
Authors
Nathan Witman, Mei Yu, Yuqi Zhang, Kexin Gai, Yuhong Chen, Lu Zhou, Christine Nguyen, Wen Zhu, Yongwei Zheng, Shawn Jobe, Mary Beth Graham, Weiguo Cui, Demin Wang, Renren Wen
Abstract
Dengue virus (DENV) vaccines should be designed to induce balanced protective immunity to all 4 DENV serotypes to mitigate the risk of vaccine-enhanced dengue disease. The first tetravalent live DENV vaccine (Dengvaxia) tested in humans was efficacious in children who were partially immune to DENV at baseline. In DENV-naive children, the vaccine was not efficacious and placed some children at risk of more severe WT DENV breakthrough infections. To define dengue vaccine responses at the individual patient level and their relationship to mild and severe dengue infections, we prospectively studied a cohort of DENV-naive children who received 1 dose of Dengvaxia. The vaccine stimulated variable responses that neutralized 0, 1 (monotypic), or 2+ (multitypic) serotypes in individual children. Using a logistic regression model, we found that vaccinated children with neutralizing antibody (NAb) to 1 serotype only were at greater risk developing dengue compared with children who were not vaccinated (odds ratio 5.07). This risk was not observed in vaccinated children with no NAb or NAb to 2 or more serotypes. We propose that individuals with durable NAb to 1 serotype have an abundance of serotype cross-reactive, nonneutralizing antibodies implicated in the enhanced replication of heterologous serotypes.
Authors
Laura J. White, Lindsay D. Hein, Maria Abad Fernandez, Cameron Adams, Elizabeth Adams, Emily Freeman, Ruby Shah, Lakshmanane Premkumar, Kristal An Agrupis, Maria Vinna Crisostomo, Jedas Veronica Daag, Michelle Ylade, Jacqueline Deen, Ana Lena Lopez, Leah Katzelnick, Aravinda M. de Silva
Abstract
Insulin and glucagon are described as having opposing actions on hepatic glycogen metabolism. However, here we showed that their coordinated action promoted glycogen turnover and meal glucose storage. In mice, pharmacological doses of insulin or glucagon failed to alter hepatic glycogen, but the combination produced a robust decrease in glycogen content. Additivity between insulin and glucagon was also seen with the activation of hepatic insulin signaling intermediates. This signaling pathway drove glycogen synthesis, suggesting concurrent actions on glycogen breakdown and repletion. A mixed nutrient meal, which stimulates an increase in both insulin and glucagon, enhanced the incorporation of dietary glucose into hepatic glycogen. This was much more pronounced than the effects of glucose alone, which only stimulated insulin secretion. These findings revealed that glucagon is required for efficient hepatic glucose storage when acting in concert with insulin. Coordinated insulin-glucagon signaling, thus, emerged as a critical mechanism for hepatic glycogen cycling, challenging the classical paradigm that these hormones work in opposition.
Authors
Nidhi Kejriwal, David Bouslov, Cheyenne R. Castle, Riya S. Karve, Galina A. Arkharova, Ashot Sargsyan, Daniel J. Drucker, Guo-Fang Zhang, David A. D’Alessio, Jonathan E. Campbell, Megan E. Capozzi
Abstract
Macrophages, endowed with remarkable phenotypic plasticity, are essential for orchestrating injury responses and regulating iron homeostasis. Given the central role of ferritin heavy chain (FtH) as a molecular rheostat linking iron sequestration to redox-dependent signaling, we examined how myeloid FtH governs renal iron trafficking and ensuing oxidative stress pathways during acute kidney injury (AKI). Transcriptome analysis revealed coupling of FtH deficiency in monocytes and macrophages with activation of ferroptosis, a regulated cell death associated with iron accumulation. Moreover, myeloid FtH deletion worsened AKI, increasing leukocyte infiltration and iron deposition, together with ferroptosis-associated gene induction, oxidative stress, and lipid peroxidation. Notably, α-synuclein (SNCA), an iron-binding protein and the main pathological driver of Parkinson’s disease, was robustly induced both by FtH deficiency and following AKI. Mechanistic studies showed that monomeric SNCA exhibits ferrireductase activity, amplifying redox cycling and promoting ferroptotic cell death. Furthermore, SNCA expression was elevated in kidney pathologies characterized by leukocyte expansion in both mouse models and human cohorts, suggesting that inflammatory microenvironments promote SNCA accumulation and redox imbalance. These findings define a macrophage FtH/SNCA regulatory axis as a key driver of ferroptosis in AKI, implicating SNCA as a pathological nexus between iron dyshomeostasis and inflammatory kidney injury.
Authors
Tanima Chatterjee, Sarah Machado, Kellen Cowen, Mary E. Miller, Bronte Johnson, Yanfeng Zhang, Laura A. Volpicelli-Daley, Lauren A. Fielding, Rudradip Pattanayak, Frida Rosenblum, László Potor, György Balla, Jozsef Balla, Christian Faul, Abolfazl Zarjou
Abstract
Although the pathogenic role of autoantibodies targeting the podocyte protein THSD7A in membranous nephropathy (MN) is well described, the consequences of autoantibody binding for podocyte homeostasis and the function of THSD7A remain unclear. Here, we induced an MN model in control and podocyte-specific Thsd7a-KO (Thsd7a–/–) mice using rabbit anti-THSD7A antibodies, followed by transcriptome and proteome analyses. Anti-THSD7A antibodies in WT mice caused significant loss of key slit diaphragm (SD) proteins, such as nephrin and NEPH1, without transcriptional downregulation. Glomeruli showed substantial transcriptomic and proteomic reconfiguration indicative of extensive podocyte injury, including disruptions in podocyte adhesion, cytoskeletal dynamics, and marked upregulation of ubiquitin-proteasome system components, cathepsins, and ADAM proteases. Notably, experiments in C3-deficient mice revealed that proteolytic activation and SD protein loss are driven by complement-independent pathways. Thsd7a–/– mice only displayed a mild phenotype under basal conditions, and they were completely protected from MN development upon anti-THSD7A antibody transfer. Finally, interactome analysis identified a protein complex, including THSD7A and integrin α3, linking THSD7A complexes to pathogenic regulation of cytoskeleton, adhesion, and membrane signaling in MN. Thus, anti-THSD7A antibodies induce profound molecular reconfiguration, including dysregulated proteolytic systems via a complement-independent pathway, revealing potential therapeutic targets in MN.
Authors
Ming Huang, Moritz Lassé, Silke Dehde, Felicitas E. Hengel, Fatih Demir, Anja M. Billing, Ning Song, Larissa Seifert, Oliver Kretz, Florian Grahammer, Ulf Panzer, Sebastian Brähler, Tobias B. Huber, Gunther Zahner, Markus M. Rinschen, Nicola M. Tomas
Abstract
Adipocytes exist along a functional spectrum: white adipocytes are energy storing, and brown adipocytes have thermogenic capacity such that activation may counteract obesity-related disease. In between are UCP1-expressing beige adipocytes, which can transition between these two energetic states. We previously showed that bone morphogenetic protein 7 (BMP7), a member of the TGF-β superfamily, enables differentiation of brown preadipocytes to mature thermogenic cells. To see whether immortalized, clonal human white and brown preadipocytes (hWAs and hBAs, respectively) would become more thermogenic in response to BMP exposure, we treated them with BMP7 or BMP4 for the first 7 days of a 30-day differentiation protocol. In hBAs, absence of either BMP7 or BMP4 led to lower expression of brown-specific markers and oxygen consumption relative to 7 days with either BMP. hWAs treated for 7 days with either BMP did not increase expression of thermogenic protein UCP1 nor induce a brown-like transcription profile. However, BMP-treated hWAs produced adipocytes that had higher basal and drug-induced maximal oxygen consumption, which was UCP1-independent and due substantially to the futile creatine cycle. Our results demonstrate that energetically quiescent hWAs can be pushed into an energy-expending phenotype without transdifferentiation into beige adipocytes, providing a new approach to treat obesity-related metabolic disease.
Authors
Kelly T. Long, Cheryl Cero, Sahara L. Ali, Nhuquynh Nguyen, Adrienne R. Guarnieri, Ju Hee Kim, Young Jae Bahn, Jurgen Heymann, Jonathan M. Dreyfuss, Sushil G. Rane, Yu-Hua Tseng, Aaron M. Cypess
Abstract
Moderate hyperoxia (30%–60% O2) in premature infants promotes bronchial airway hyperresponsiveness (AHR) via airway smooth muscle (ASM), a key regulator of bronchoconstriction, bronchodilation, and remodeling. Understanding how O2 exposure drives long-term bronchial changes in prematurity is critical for developing therapies for airway disease across the lifespan. Premature lungs have immature antioxidant defenses, potentially due to disrupted mitochondrial dynamics, increasing susceptibility to O2-induced oxidative stress. Thus, mitochondrial homeostasis is highly relevant to ASM dysfunction and airway disease. We propose that hyperoxia in prematurity promotes mitochondrial dysfunction, and that the gasotransmitter hydrogen sulfide (H2S) mitigates O2-induced mitochondrial damage in developing ASM. Human fetal ASM (fASM) cells were exposed to moderate hyperoxia to investigate the effects of exogenous H2S donors (GYY4137, AP39) and stabilization of cystathionine β-synthase (CBS), an H2S biosynthetic enzyme, on mitochondrial structure and function. Hyperoxia impaired fASM cell mitochondrial integrity, while H2S donors in particular, or CBS stabilization attenuated adverse O2 effects on mitochondrial morphology, ROS, respiration, calcium regulation, and contractility. These findings highlight the therapeutic potential of H2S in the premature lung exposed to moderate hyperoxia.
Authors
Colleen M. Bartman, Michael Thompson, Samantha K. Hamrick, Niyati A. Borkar, Daniel Pfeffer-Kleemann, Preetham Ravi, Marta Schiliro, Yak Nak, Christian Vivar Ramon, Li Drake, Y.S. Prakash, Christina Pabelick
Abstract
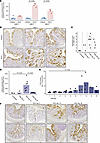
Heterogeneity in disease severity and treatment response in inflammatory bowel disease (IBD) likely evolves from individual differences in host-microbiota-immune interactions. Histological evaluation of intestinal biopsies is central to diagnosis, but histological parameters that define underlying immune mechanisms are limited. We investigated histological features that distinguish individual patient immune profiles in therapy-naive pediatric IBD patients (age 6–18 years) using biopsy immunohistochemistry and transcriptomics and plasma proteomics across two cohorts. High colonic epithelial expression of secretory leukocyte protease inhibitor (SLPI), a microbiota-induced regulator of epithelial function, occurred in IBD patients with high clinical disease activity and more severe endoscopic and microscopic disease activity. SLPI expression was related to increased neutrophil infiltration, transcriptomic signatures of activation, and genes known to associate with therapeutic resistance. High SLPI colocalized with high densities of IL-17–secreting cells and was associated with high plasma concentrations of Th17-related immune proteins. Additionally, patients with high intestinal SLPI had an intrinsically different immunotype, in which circulating neutrophils exhibited altered transcription of genes involved in neutrophil granule formation, phagocytosis, oxidative phosphorylation, and interferon signaling. Thus, high colonic SLPI expression at diagnosis associates with severe IBD, increased IL-17A–neutrophil pathway responses, and altered transcriptomic wiring of circulating neutrophils.
Authors
Sandrine Nugteren, Beatriz Calado, Ytje Simons-Oosterhuis, Daniëlle H. Hulleman-van Haaften, Willem K. Smits, Renz C.W. Klomberg, Bastiaan Tuk, Mohammed Charrout, Dicky J. Lindenbergh-Kortleve, Michail Doukas, Mathijs A. Sanders, Gregory van Beek, Johanna C. Escher, Lissy de Ridder, Maria Fernanda Pascutti, Janneke N. Samsom
No posts were found with this tag.